X-ray imaging is a low-cost, powerful technology that has been extensively used in many industries, including medicine, manufacturing, security, and astronomy.
X-rays were first discovered in 1895 by German physicist Wilhelm Conrad Roentgen, when he was conducting experiments with a cathode-ray tube consisting of a glass bulb encapsulating positive and negative electrodes in a vacuum covered in heavy black paper. He noticed that when he applied a current, mysterious light rays emitted from the bulb that could pass through most materials, including human tissue. Wilhelm called them “X”, the mathematical symbol for an unknown variable.
This momentous discovery revolutionized the fields of physics and medicine. X-rays took the world by storm and within months, went from laboratory experiment to clinical use with medical radiography machines being built. The first angiography, moving-picture x-rays, and military radiology were performed in 1896. Roentgen was awarded the first Nobel Prize in Physics in 1901.
Over the past century, X-ray technology has continued to evolve, primarily driven by advancements in detection and imaging technology. In the 1950s and 1960s computerized axial tomography (CAT) scans were developed, which allowed doctors to create detailed, three-dimensional images of the body. In recent years, there has been renewed interest in X-ray technology, driven in part by the growing field of medical imaging startups and AI. However, the method for creating X-rays has remained fundamentally the same as when it was discovered, the X-ray tube. But new types of X-ray sources on the horizon promise to create new possibilities for the future of X-ray imaging.
Forced to choose: Low-dose or image quality
X-rays have very short wavelengths and a high energy level that allow them to penetrate materials that light cannot, such as the human body, thus making them a valuable tool for medical imaging. However, the high energy of the X-rays causes chemical bonds in the body to break, and excessive x-ray exposure can cause tissue damage and increase the risk of developing cancer.
To achieve a high resolution and contrast in diagnostic imaging, applications such as computed tomography (CT) scans for angiograms or colonoscopy require high x-ray doses. A 2009 study estimated that the overall lifetime risk of cancer increased 4-17% in patients who had multiple CT scans across a span of about 20 years. These consequences limit the utility of current x-ray imaging as a screening tool for at-risk patients for various diseases. Hence, any advanced X-ray technologies would need to lower radiation exposure risk in order for them to be adopted by medical providers.
An X-ray imaging system basically comprises an X-ray source and an X-ray imaging detector. We explore some of the latest advances made in these two areas to lower dosage while improving X-ray image quality.
Digital Radiography and next generation x-ray detectors
A major leap in X-ray imaging came in the 1980s with the advent of digital radiography, which produces a digital radiographic image instantly on a computer using x-ray sensitive plates to capture data without the use of a film or cassette (analog). This also led to the development of computerized axial tomography (CAT) scans which allowed doctors to create detailed three-dimensional images of the body by taking multiple images at different angles.
Digital radiology is the most popular form of imaging today due to its seamless integration with the digital world. Images can be reviewed quickly in real time allowing faster processing, improved image quality with better signal-to-noise ratio without increasing radiation exposure, improved dynamic range for higher sensitivity and digital enhancement with AI. Additionally, the use of digital radiography reduces the need for retakes, which also helps lowers radiation exposure.
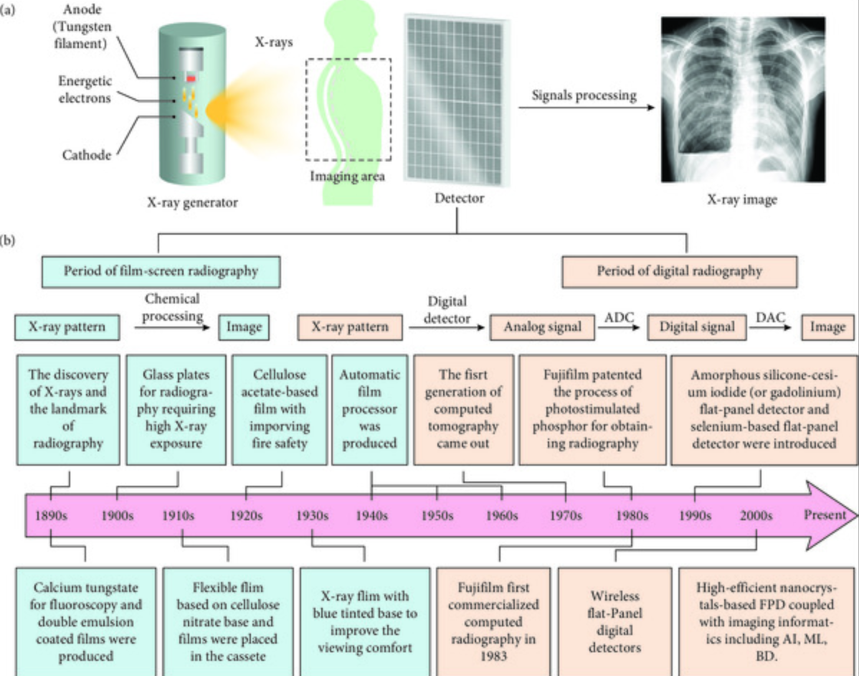
Development of X-ray radiography and the evolution of x-ray detectors. Image source: https://spj.science.org/doi/10.34133/2021/9892152
This leap from analog to digital was made possible by advancements in detector technology and the invention of flat-panel detectors (FPDs), or digital detector arrays. The most advanced FPDs today convert incident X-ray radiation into an equivalent electric charge and then to a digital image through a detector sensor. Most current flat panel detectors use Cadmium zinc telluride (CZT) or amorphous selenium (a-Se) on an electrode plate. X-ray photons are directly converted into digitized and amplified electronic signals when they impact the Cadmium telluride or amorphous selenium. Thin film transistors are used to read the data on the detectors.
The latest breakthroughs in detector technology are photon counting X-ray FPDs that are extremely sensitive, with the ability to resolve even individual photons of incident X-rays. This technology has enabled higher contrast-to-noise ratio, improved spectral and spatial resolution and the ability to lower the X-ray dose by requiring less X-ray intensity.
Global giants in the medical imaging technology space are racing to develop the first Photon Counting CT (PCCT) scanner. General Electric (GE) Healthcare acquired Swedish start-up, Prismatic Sensors, which specialized in developing photon counting detectors and more recently launched their Deep Silicon technology. Canon Medical Systems also acquired a Canadian CZT detector company, Redlen Technologies, to accelerate the development of their own PCCT machine.
Photon counting detector technology has enabled the development of spectral X-ray imaging, which can measure and determine material composition such that one can distinguish bone, fat and water and produce 3D color X-ray images. This is being developed and commercialized by MARS Bioimaging, based on technology developed at CERN, the European Organization of Nuclear Research. The company has a commercial device for research use in the laboratory and are developing a medical grade device.
Rapid advances in materials sciences also promise to revolutionize X-ray detector technology. Perovskites, a superstar semi-conductor that is being developed for use in high-efficiency solar cells, also show great promise in X-ray detection due to its ability to strongly absorb X-rays. Compared to commercial FPDs using amorphous selenide, perovskites are lower-cost, have higher sensitivity (allowing for half the dose or double the resolution) and easier to synthesize. Recognizing the promise this material holds, the EU-funded the PEROXIS project which brings together a consortium of academic and industrial players to develop perovskite-based detectors. Major x-ray imager manufacturers, like Samsung, and startup companies, like Actinia, have also started developing perovskite detectors, but most are still early in development.
Reinventing the X-ray source
To date, most advances in X-ray imaging have been made on the detector side. The X-ray source used in all commercial x-ray machines still rely on the same X-ray tube, where X-rays are produced by bombarding a metal target with electrons, that was developed back in 1895. However, the simplicity and low cost of this technology has enabled it to endure.
The shortcomings of X-ray tubes are
that the light produced is quite messy – it is made up of many frequencies (colors), incoherent, are relatively low in intensity and because the light source is large, it limits the resolution they can achieve. An X-ray source that overcomes these limitations would spur a new wave of applications for X-ray imaging.
In recent years, there has been a drive to adapt the X-ray tube to be able to achieve higher resolutions by making the light source smaller. Such devices, called microfocus X-ray sources, have helped unlock applications in precision industrial imaging, where with the advent of additive manufacturing requires manufactured products to be imaged at the micron scale during production. But the low intensity, low X-ray energy and poor lifetime of such devices has limited their wider commercial use.
Next generation X-ray sources with all the desired properties do exist but are today at stadium-sized accelerator facilities called synchrotrons. These are the brightest and most advanced X-ray sources available, producing X-rays by accelerating electrons to nearly the speed of light and driving them through a ring lined with powerful magnets to generate high-energy monochromatic X-rays. These X-rays enable new imaging modalities with nanoscale detailed information. However, there are only ~70 such facilities around the world, each costing hundreds of million to build, with use restricted to research and development applications and long waiting times for their use.
A miniaturized version of synchrotrons has been in development since the 1970’s called the free-electron laser (FEL). But these devices are the size of a house and despite years of development, the quality of light produced remains poorer than synchrotrons and so have not found commercial use.
To overcome this scalability problem and enable widespread deployment of synchrotron-grade X-rays, Lumitron Technologies, a company based in Irvine California, has developed a new-type of X-ray source called a laser-Compton source. Their technology recreates the power of a synchrotron in a device that is the size and cost of a modern MRI scanner. Lumitron’s imaging device, the HyperView, can reduce radiation by 100-fold or increase resolution by 1000-fold. Their proprietary technology is built on 17 years and US$220 million worth of federally funded R&D from Lawrence Livermore Labs where Lumitron’s founder, Dr. Chris Barty, was Chief Technology Officer of the world’s largest laser facility, that was recently used to demonstrate net energy gain in a high-profile breakthrough for the nuclear fusion field.
Because the Lumitron device has 100 times less radiation dose, using X-ray imaging as a regular screening tool to detect cancers becomes a possibility. Furthermore, the technology enables theranostics, which is the ability to use X-rays to both diagnose as well as treat at the same time. This works by tuning wavelengths of X-rays to activate specific elements on therapeutic nanoparticles that home in and target the cancer cells. Imagine a future where a doctor will be able to make a cancer diagnosis during an imaging session and without moving the patient, use x-rays to zap the cancer with pinpoint accuracy at the same time.
However, we are just starting to scratch the surface of possibilities that will open when an advanced X-ray source becomes widely available in labs and clinics. For example, phase-contrast X-ray imaging that would allow imaging of soft-tissue with an X-ray, have been researched for years. But the lack of bright, coherent X-ray sources from a small light source has hindered development. A recent upgrade at a synchrotron facility has enabled a study called Hierarchical Phase-Contrast Tomography (HiP-CT) which has imaged whole organs at a resolution down to 1 micron (100x that of conventional CT scans) and captured the imagination of the medical imaging community. This unprecedented detail in structural information at the point of care would improve insights in clinical imaging and aid better diagnosis.
HiP-CT would remain a niche modality if it can only be done at synchrotrons. Lumitron’s HyperView would enable and democratize access to HiP-CT as their machines are the size and cost of an MRI machine. And with such technology, a future with 3D mammograms of extraordinary detail without limitations in breast size or the need for breast compression during imaging can be enabled.
With better detectors, new x-ray sources and ever-increasing advancements in computer capabilities, improved X-ray imaging systems hold great promise to improve human health and lives.



